Syncora
Designed for efficient study startups, Syncora offers a centralized platform that enhances real-time data management and stakeholder collaboration. It automates repetitive tasks, streamlines workflows, and provides real-time analytics, reducing errors while ensuring regulatory compliance. With robust encryption and customizable themes, it simplifies the initiation process, allowing researchers to focus on essential scientific work.
Top Syncora Alternatives

StackScan
Identify and analyze websites by their tech stack with access to 50,000+ technologies and a database of 105 million domains.
StudyProtocol
Designed for clinical trial management, StudyProtocol allows research teams to effortlessly oversee their studies.
MyDataHelps
Designed for modern clinical research, this Clinical Trial Management Software streamlines digital trials by integrating real-world data through a no-code platform.
Ripple Science
Designed for clinical research teams, Ripple Science streamlines patient recruitment and management through an intuitive, web-based platform.
Intrinsic CTMS
Designed for optimal efficiency, Intrinsic CTMS empowers research teams to seamlessly plan, manage, and monitor their clinical studies.
Curebase
Curebase offers cutting-edge eClinical software powered by AI, streamlining study launches and enhancing data quality.
SimpleTrials
Designed for Sponsors, CROs, and Sites, this clinical trial management system offers a flexible and cost-effective solution starting at $599/month without long-term commitments.
Cloudbyz Safety and Pharmacovigilance (PV)
It streamlines drug safety operations by automating case intake and regulatory reporting, enabling faster decision-making...
Jeeva eClinical Cloud
It supports real-time data collection, remote patient engagement, and efficient scheduling, allowing researchers to adjust...
Cloudbyz eTMF
It provides real-time visibility and ensures compliance, streamlining collaboration among sponsors, CROs, and monitors while...
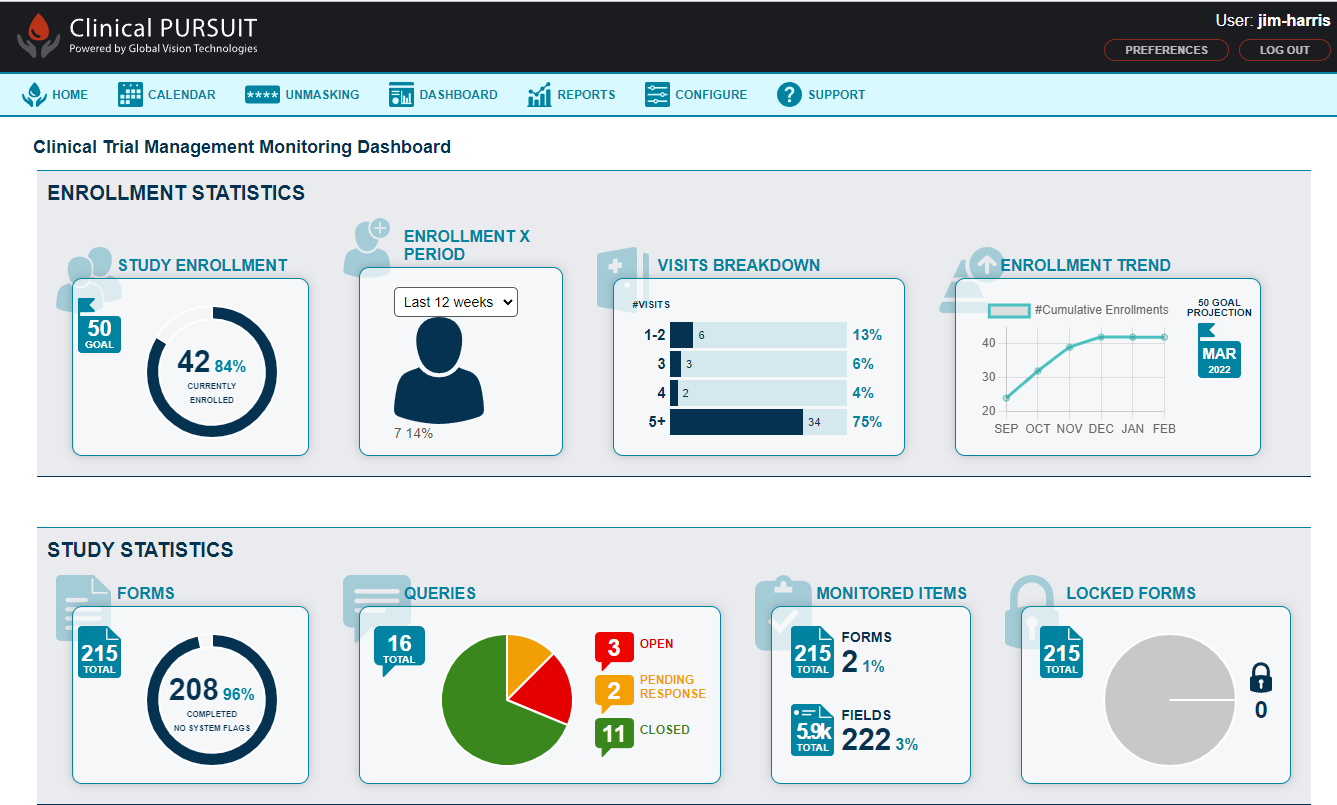
ClinicalPURSUIT
By utilizing a web-based solution, it significantly reduces study costs while ensuring secure data management...
Clinical Study Insight
It offers mobile apps for iOS and Android™, enhancing engagement through reminders and tailored content...
Clinical StudyPal
It facilitates efficient patient recruitment, real-time communication, and comprehensive data collection through user-friendly apps and...
Clindata Cloud
It automates the generation of submission-ready CDISC datasets and analytics, ensuring compliance with FDA standards...
Datacubed Health
Its adaptable application ensures participants easily understand protocols while maintaining active involvement through a proprietary...
ClinAccess
By eliminating the need for data transfer from Oracle/SQL, it enhances productivity...
Top Syncora Features
- Real-time data access
- Automated reporting tools
- Centralized site coordination
- Customizable dashboard themes
- Seamless team communication
- Efficient protocol management
- Comprehensive audit trails
- Robust data encryption
- Adaptable study parameters
- Integrated workflow automation
- Quick study startup processes
- Free support and updates
- Detailed compliance reporting
- Streamlined document handling
- Rapid participant data management
- Diverse scenario accommodation
- Minimized manual data entry
- Enhanced transparency measures
- Timely study initiation tracking
- User-friendly interface design.